Tag: chemistry
Clean Energy Project 2
My friends at the Clean Energy Project have started a second stage that focuses on quantum chemistry calculation. You might recall [previously: here and here] that the Clean Energy Project consists of screen saver that allows you to donate computer time to do calculations to discover better materials for solar energy.
—
Time flies like an arrow. Fruit flies like a banana.
-Groucho Marx
Chemical Party
Xe is lonely.
—
Enoch shrugs modestly. “Where I grew up, memorizing the digits of pi was the closest thing we had to entertainment.”
-Cryptopnomicon
What is Time-Dependent Density Functional Theory?
Time-Dependent Density Functional Theory looked like a mess when it was first explained to me. I probably made the face of a person who smells sushi for their first time, wondering if this is some sort of bad joke.
After all, quantum mechanics is supposed to be a theory of non-commuting observables that evolve in a linear fashion. TD-DFT is nothing like that, yet it claims to reproduce all the same effects. Fishy indeed.
TD-DFT first focuses on the density of the wave function, in particular, the position basis of it. This is relevant for chemical calculations where it is very important to know where are the electrons. Of course, the density is one of many observables that are relevant, but TD-DFT makes it stand out by letting this observable evolve by means of a functional of itself. In other words, you don’t fully evolve the wave function by means of an operator, but instead you have a very complicated, non-linear functional that takes as its input the density and lets it evolve. In practice, since the functional is non-linear, in practice, the evolution is done iteratively.
Runge and Gross proved that if you only cared about the evolution of one observable, the density, this procedure is equivalent to the full quantum mechanical evolution. In other words, you can map the evolution of a particular observable the wavefunction under Schrodinger’s equation into a functional of the same observable.
What you gain from this approach is a computational speedup. The prize paid is that writing the exact functional is actually a very hard problem, at least as hard as doing the full quantum mechanical evolution. However, in practice, approximated functional can be written down and used for real calculations that can predict properties for real materials. This technique is widely used, mostly as a black box toolkit used by many physical chemists around the world.
In our latest papers, we were able to show that this mapping can be also performed for open quantum systems instead of just Schrodinger’s equation. First, we developed the general theory of how the Runge-Gross theorem can be generalized, placing it in context of previous incomplete attempts. This paper was published in PCCP as a Hot Article. In it, we discuss how the theorem works even in the highly non-Markovian regime of an open quantum system.
In our second paper, we take this even further. The evolution of an observable of an open quantum system can be mapped to a functional for a close system. At first, this seemed counter-intuitive. After all, you cannot map the evolution of an open system into a closed system.
O te peinas o te haces rolos.
However, if you only care about one observable, and you are willing to use non-linear functionals, this can be done consistently, for just that observable. Since most of the code written for TD-DFT was for closed systems, our results shows that those techniques could be used to model open quantum systems. We feel that new chemical calculations with thermodynamic effects can now be explored with this theory.
—
Dr. Strangelove: It is not only possible, it is essential.
Our paper chosen as PCCP Hot Article!
Our paper
Time-dependent current-density functional theory for generalized open quantum system
Joel Yuen-Zhou, César Rodríguez-Rosario and Alán Aspuru-Guzik
In this article, we prove the one-to-one correspondence between vector potentials and particle and current densities in the context of master equations with arbitrary memory kernels, therefore extending time-dependent current-density functional theory (TD-CDFT) to the domain of generalized many-body open quantum systems (OQS). We also analyse the issue of A-representability for the Kohn–Sham (KS) scheme proposed by D
Agosta and Di Ventra for Markovian OQS [Phys. Rev. Lett. 2007, 98, 226403] and discuss its domain of validity. We suggest ways to expand their scheme, but also propose a novel KS scheme where the auxiliary system is both closed and non-interacting. This scheme is tested numerically with a model system, and several considerations for the future development of functionals are indicated. Our results formalize the possibility of practising TD-CDFT in OQS, hence expanding the applicability of the theory to non-Hamiltonian evolutions.
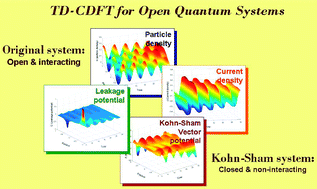
has been published on PCCP and chosen as a Hot Article !
SQuInT, APS, Publications
I’m back.
After a long, tough, wintery and busy month, I’m back.
Progress report follows.
Alright, first, I went to SQuInT. The Southwestern Quantum Information and Technology conference isn’t true to its name. It was held in the Northwest, Seattle, where beautiful weather seemed to tunnel through the mountains’ potential just for us. The conference itself was very productive and I had the opportunity to see family, friends and collaborators.
In other news, we submitted a paper on Open Quantum Systems and Time Dependent Current Density Functional Theory titled Time-dependent current-density functional theory for generalized open quantum systems to the journal Physical Chemistry, Chemical Physics (PCCP). It has been accepted for publication and might appear in a special issue on Time Dependent Density Functional Theory.
We also submitted a related paper to another journal, paper titled Time-Dependent Density Functional Theory for Open Quantum Systems using Closed Systems. You can read it in the arXiv.
Finally, I went to the APS March Meeting, where 7,000 physicists took over the city of Pittsburgh, where I was able to find bars decorated with Roberto Clemente posters, where a restaurant served Carrucho (Conch). Sometimes I feel the APS March meeting is too big, too overwhelming, talks are too short, and there is too much going on simultaneously. But then I’m surprised by meeting people I hadn’t seen in almost 10 years now, and by how APS March meeting always lead to new collaborations.
Exciting times these are.
—
“I have wept three times in my life. Once when my first opera failed. Once again, the first time I heard Paganini play the violin. And once when a truffled turkey fell overboard at a boating picnic.”
-Gioachino Rossini
Outreach: Back to La Iupi
I just came back from Puerto Rico, into the Bostonian winter.
Although I had a very brief opportunity to see my people back home, the main purpose of the trip was work. Several young professors from the Chemistry and Chemical Biology department at Harvard have started an effort to outreach, but for real. The main purpose of the program is to visit universities with a big population of under-represented minorities. I proposed to go to Universidad de Puerto Rico, Rio Piedras. I still have many friends there, and it was very good to see again so many professors that gave me many opportunities as an undergrad to grow as a researcher. Without their support, I wouldn’t be a physicist now.
The outreach activity was very successful. The people at the Resource Center for Science and Engineering did an amazing job of coordinating the activity. We were able to talk to many researchers there, and spent a good day with many talented science undergraduate students from all around the island. I’m sure new collaborations will grow from this.
To see information about many undergraduate research programs and how to apply, click here.
Donate computing time for the environment
There is the environmental need and the political will to take solar energy seriously. Our group at Harvard is leading several theoretical and computational efforts to develop more efficient solar panels. One of our efforts (not my project) is to use computational chemistry tools to find novel materials that would lead to better solar technologies. This is mostly performed by trial and error. A lot of it.
A certain molecular arrangement is “proposed” randomly, and the computer calculates its molecular energy to see if it makes sense (if it is a realistic material) and then if it is useful for solar panels. This trial and error approach takes a long time, requires a lot of computational power, but it can be parallelized in a straight-forward manner.
How can you help us?
By downloading the Harvard Clean Energy Project software. With it, you can donate your unused computer cycles, when the computer is on but not using the processor much, to help perform the combinatorial calculations. With these small computing contributions from thousands of students it is expected that the calculations will be done ten times faster than in a supercomputer.
Right now it is only available for Windows, but in the next few weeks it should be compatible with Linux and Mac too.
The project has had a lot of visibility, the other day BBC called our office!
Imagine a molecule. Now Imagine a quantum computer solving it.
So, we have the periodic table. We know that atoms combine into molecules depending on their energy spectrum, its energy levels. We know quantum physics, the theory that reigns in the atomic regime.We know math. We know quite a lot, actually.
So, we want to create new chemicals. Having new materials would give us new technologies, having new molecules would provide us with new medicines, saving millions of lives.
How come it is so hard to use what we know to get what we want?
The problem is that atoms have many electrons, and you have to calculate the equations for each electron. But, electrons interact with other, the solution of the equations of one depend on the solutions of the equations of the other. The solutions are interconnected, coupled. This is know as the many body problem. This makes solving the equations very hard.
Although we know what to do to calculate the energy of a complicated molecule, we can’t actually do it. It takes too long, even for a computer. Computers get faster every year, but they don’t get fast fast enough for the problem. Making the molecule just a bit more complicated demands us to have a computer much much much more powerful than for one a bit simpler. In other words, the problem of solving the energy of a molecule doesn’t “scale” well.
Unlike a conventional computer, Aspuru-Guzik and his colleagues say, a quantum computer could complete the steps necessary to simulate a chemical reaction in a time that doesn’t increase exponentially with the reaction’s complexity.
What is a quantum computer? How is it different from other computers? What tricks can it do to solve chemical reactions faster than a normal computer?
This blog is about those questions and more. Stay tuned.

